Home
News
IMB-CNM consolidates its position as the second center in Catalonia for spin-off investment over the past decade
The creation of highly resilient dosimeters for FLASH radiotherapy and the ability to mobilize € 124 million in investment through its spin‑offs reinforces the institute’s leading role in innovation and technology transfer in Catalonia
Scientific referents in our community for a new February 11
The IMB-CNM is focusing its activities for the International Day of Women and Girls in Science on a public campaign to show the profile of its female researchers. In schools, informative talks have been coordinated in five different locations
Agenda
Highlights

Accurate neutron detection in mixed photon-neutron and pulsed radiation fields is technically challenging, impacting industrial and medical applications. This paper presents the first measurements of thermal neutrons in conventional radiotherapy accelerators using a silicon carbide (SiC) P–N diode with different neutron converters. SiC detectors enable real-time estimation of secondary thermal neutron contributions, crucial for emerging radiotherapy techniques requiring precise neutron fluence monitoring. Beyond medical applications, the presented detectors show potential for neutron dosimetry, radiation monitoring, nuclear safety, and scientific research. The SiC diode active detection layer is less than 30 µm thick, and provides excellent gamma rejection, allowing discrimination of neutrons-induced events in mixed radiation fields. Experimental tests conducted on a TrueBeam radiotherapy LINAC demonstrated a thermal neutron detection efficiency of (4.32 ± 0.02)% for a (50 ± 10) µm thick LiF neutron converter. The detector, placed at 1.2 m from the accelerator isocenter, was used to measure neutron fluences at different monitor unit (MU) rates, ranging from 100 to 600 MU/min, with the LINAC operating at 15 MV. Under these conditions, the detector exhibited good linearity, without saturation or dead time effects.
Sci Rep 15, 30543 (2025). https://doi.org/10.1038/s41598-025-13052-w
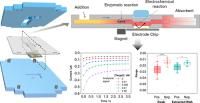
Providing viral load numbers of infection events aids in the identification of disease severity and in the effective overall patient management. Gold-standard polymerase chain reaction (PCR) techniques make this possible but cannot be applied at the point of need and in low-resource settings. Here, we report on the development of a compact analytical platform that can detect a conserved sequence of the RNA of severe acute respiratory syndrome-coronavirus 2 (SARS-CoV-2) in 40 min in nasopharyngeal swab samples without the need for any previous purification or gene amplification steps. It combines electrochemical and paper fluidic approaches together with a sandwich hybridization assay performed on magnetic nanoparticles (MNPs) modified with a tailor-designed capture DNA hairpin. The device proves to quantitatively detect viral RNA in a retrospective study carried out with nasopharyngeal swab samples. A sensitivity of 100% and a specificity of 93% were estimated by the receiver operating characteristic (ROC) analysis. However, although molar concentration values of the target RNA sequence are provided, these estimates do not fully correlate with the viral load numbers estimated by RT-qPCR over the whole Ct sample range. Empirical studies have been carried out that have provided clear insights into this hurdle and simple solutions to overcome it, without depriving the device of the features required for potential use in a point-of-care (PoC) environment.
Anal. Chem. 2025, 97, 22, 11863–11873. DOI: 10.1021/acs.analchem.5c01605